Carbon Nanotube Technology for Robust Desalination Membranes
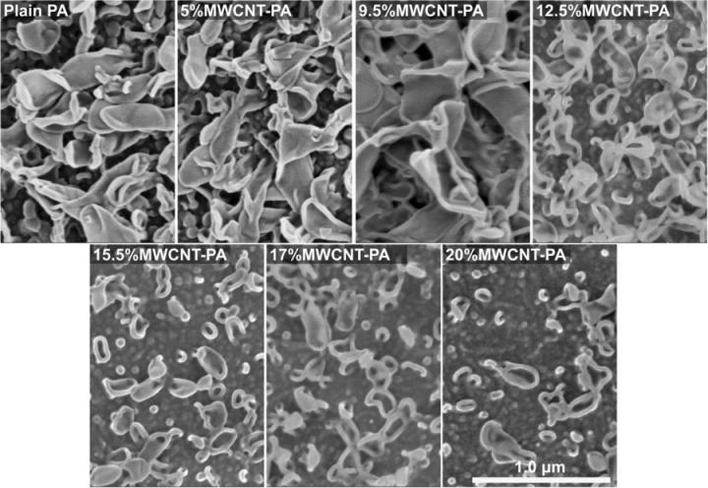
SEM images of MWCNT-PA (Multi-Walled Carbon Nanotube-Polyamide) nanocomposite membranes, for plain PA, and PA with 5, 9.5, 12.5, 15.5, 17 and 20 wt.% of MWCNT, where the typical lobe-like structures appear at the surface. Note the tendency towards a flatter membrane surface as the content of MWCNT increases. Scale bar corresponds to 1.0μm for all the micrographs. (Credit: Copyright 2018, Springer Nature, Licensed under CC BY 4.0, https://www.eurekalert.org/multimedia/pub/167626.php?from=390445)
Researchers from Shinshu University in Japan have used carbon nanotubes to develop robust reverse osmosis membranes that can withstand water desalination on a large scale. This technology may be able to improve efficiency and reduce the costs of desalination in a world experiencing a growing need for clean water. Morinobu Endo, a distinguished professor at Shinshu University and the Institute of Carbon Science and Technology‘s Honorary Director, corresponded with EM about this research.
Desalination and reverse osmosis technology
Although researchers have been developing reverse osmosis (RO) membrane technology for decades, our world is changing rapidly. Novel threats such as stressed urban water supplies and climate change challenge conventional water treatment and supply systems.
Thin film composite systems are what typically make up reverse osmosis membranes, and these use an active layer of polymer film to stop unwanted substances, including salt, from moving through a permeable substrate. These membranes can produce water for irrigation or drinking, but they are expensive, with regard to both financial cost and energy use. Dr. Endo and the team are applying nanotechnology to this problem.
“Our group has a recognized expertise in carbon nanostructures such as carbon nanotubes (CNT), which we successfully used for developing reverse osmosis (RO) membranes for improved water desalination processes,” explains Dr. Endo. “Our motivation was to overcome some of the drawbacks of the conventional membrane technology, such as the robustness against chlorine degradation and propensity to fouling. We already know that CNTs often improve the chemical resistance of polymers, and recently we found they are also excellent additives for polyamide (PA) RO membranes.”
Keeping a technology like desalination affordable means that membranes must be robust in terms of efficiency of separation, and stable, withstanding the harsh conditions cleaning treatments create. To solve the problem, Endo and his team developed a multi-walled nanocomposite membrane composed of polyamide and carbon nanotubes, which resists chlorine. Degradation caused by chlorine is among the main causes of RO membrane failures, but the additional carbon nanotubes stabilize the polyamide’s linked molecules, protecting the membrane against chlorine.
This RO membrane purifies water, removing other contaminants, not just salt.
“The addition of CNT does not negatively affect the rejection of salt and other contaminants,” details Dr. Endo. “The PA-based RO membrane technology is widely used as the most efficient way to remove dissolved ions in water. Since the RO principle does not change for our CNT nanocomposite membranes, they are able to remove all the other contaminants from water that conventional RO membranes do.”

AFM images of MWCNT-PA nanocomposite membranes. The 3D views are shown for samples before (left) and after (right) chlorine exposure. From top to bottom the rows show samples of plain PA, 9.5 wt.% and 20 wt.% MWCNT-PA for analysis. The average roughness (R a ) exhibits the different degradation behavior as a function of MWCNT content within the nanocomposite membranes. (Credit: Copyright 2018, Springer Nature, Licensed under CC BY 4.0, https://www.nature.com/articles/s41598-018-21192-5/figures/2)
Scaling up, moving forward
The team is now working to expand and stabilize the processing and production of these multi-walled carbon nanotube-polyamide nanocomposite membranes.
“There are several key steps within the whole nanocomposite membrane fabrication process, such as CNT dispersion, active layer adhesion to support membrane, post-fabrication surface treatment, etc., which become very critical when moving from laboratory scale membrane production to pilot scale membrane production,” Dr. Endo states. “These steps need to be carefully engineered for continuous membrane production.”
Scaling up the method of synthesis will start with pilot plants.
“We would set up pilot plants for nanocomposite membrane fabrication, where each step of our original process developed in lab is being implemented in large scale,” Dr. Endo describes. “The membrane fabrication, which in the starting research phase was done manually, is being implemented by an automated and continuous process, as conventional RO polymeric membranes are produced. These larger membranes are obtaining similar filtration performance as the smaller ones made in the laboratory.”
The team also plans to collaborate with others to produce commercial membranes based on their new technology. The Japan Science and Technology Agency supports the research and is now supporting the team into the scaling-up phase. “According to our roadmap, the project is progressing,” adds Dr. Endo.
This nanocomposite membrane resists chlorine, and then removes the dissolved chlorine ions from the water during the RO process, as typical polymeric RO membranes do. The material can also be adjusted to remove other kinds of molecules.
“We are working to adjust the composition in our nanocomposite membrane in order to efficiently remove boron, which is difficult to separate,” remarks Dr. Endo. “Boron in irrigation water can be harmful for some crops. In the case of other molecules, such as dyes and complex chemicals, RO membranes can remove them efficiently, given that the sizes of such contaminant molecules are larger than the pores in RO membranes, making their passage extremely difficult.”
Moving forward, Dr. Endo and the team are going to improve and continue to develop the membrane.
“The incorporation of CNT to any polymeric matrix to form a nanocomposite is not an easy task, given the many factors such as CNT properties, dispersion fabrication, and membrane methodology that need to be tuned,” states Dr. Endo. “We are constantly improving all the conditions to obtain better membranes.”
Top image: SEM images of MWCNT-PA (Multi-Walled Carbon Nanotube-Polyamide) nanocomposite membranes, for plain PA, and PA with 5, 9.5, 12.5, 15.5, 17 and 20 wt.% of MWCNT, where the typical lobe-like structures appear at the surface. Note the tendency towards a flatter membrane surface as the content of MWCNT increases. Scale bar corresponds to 1.0μm for all the micrographs. (Credit: Copyright 2018, Springer Nature, Licensed under CC BY 4.0, https://www.eurekalert.org/multimedia/pub/167626.php?from=390445)











jarboes
April 7, 2022 at 4:17 AM
Absolutely! A reverse osmosis system is one of the most extensive methods of filtration. It removes 98% of dissolved solids, which makes it healthier to drink. A water distiller is the only other drinking water system that also reduces TDS, but it’s less efficient than an RO system.