USGS Scientists Identify Causes of High Concentrations of Radium in Aquifer Water
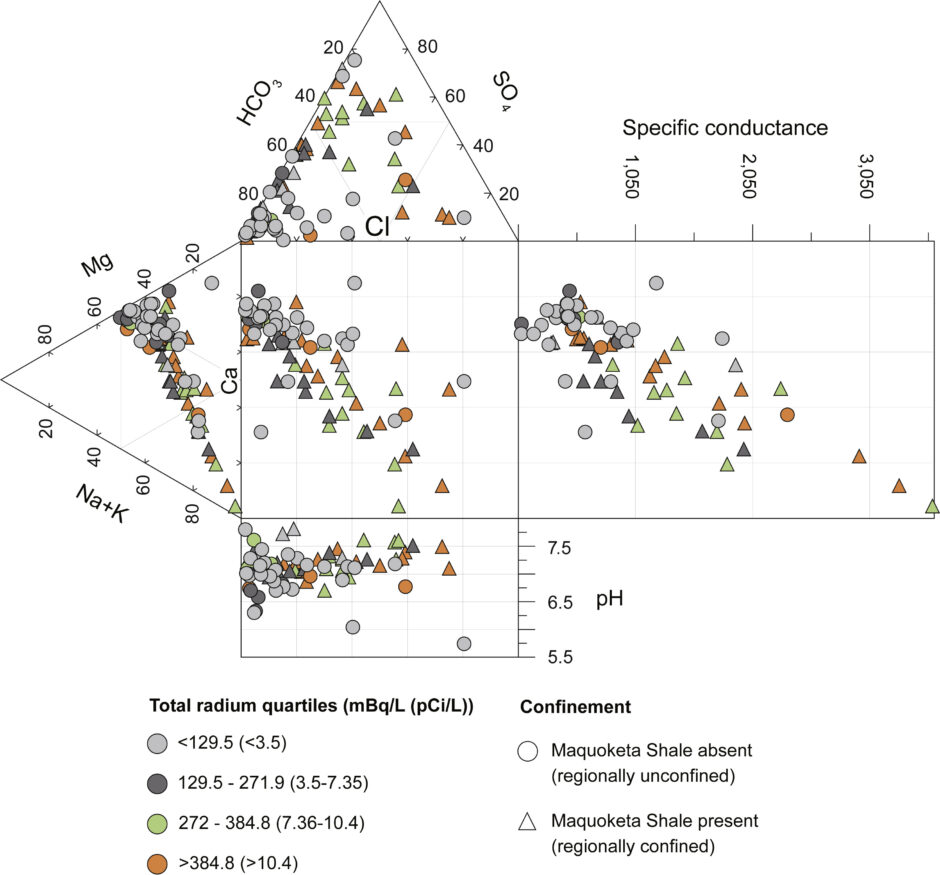
Water type becomes increasingly mineralized (SO4 or Cl dominated) and total Ra increases where the C-O aquifer system is regionally confined by the Maquoketa Shale. (Credit: Authors, USGS)
What exactly is happening far beneath our feet is typically a bit mysterious, requiring some special effort to study. Starting in the 1950s, reports of radium concentrations in excess of 5 picocuries per liter (pCi/L) in water from the Cambrian-Ordovician aquifer system started scientists thinking about the issue of radium in this massive aquifer which provides more than 630 million gallons of water each day to the public supply in Iowa, Illinois, Minnesota, Michigan, Wisconsin, and Missouri. Now, USGS scientists have published new research results, available online here, revealing how much radium is in the aquifer, and shedding light on how it gets there.
Radium in the Cambrian-Ordovician aquifer system
Paul Stackelberg, a hydrologist in the New York Water Science Center of the United States Geological Survey (USGS), explains the details that the team was working with.
“The U.S. Environmental Protection Agency (U.S. EPA) has set a maximum contaminant level (MCL) of 5 pCi/L for the summed concentration of two radium isotopes (Ra-226 + Ra-228) in public-drinking-water supplies,” Stackelberg explains. “Since at least the 1950s, there have been reports of radium concentrations in excess of 5 picocuries per liter (pCi/L) in water from the Cambrian-Ordovician aquifer system. Most of these previous studies, however, were based on radium measurements from individual wells or from studies that were local to regional in scale. Our study was conducted as part of the USGS’s National Water Quality Assessment (NAWQA) project and is the first to systematically access the quality of water across all potable portions of the Cambrian-Ordovician aquifer system.”
It’s important to know where radium is present in groundwater—and how much is there—because of the health risks that come with drinking water that has high levels of radioactive isotopes in it.
“Studies have been published linking increased rates of leukemia, bone cancer and other adverse health effects to drinking-water supplies with elevated levels of radium,” details Stackelberg. “These studies, however, often do not report on the source(s) of the drinking water. Several studies have been conducted in the Midwestern USA, but they too did not specifically identify the source of the drinking-water supplies.”
Obviously, then, a large part of what the USGS needed to do was identify how much of a risk is posed to the public by radium in the aquifer’s water. The team dealt with alpha-particle emitting Ra isotopes 224Ra and 226Ra by separating, purifying, and mounting them on a source, and then counting them using alpha-spectrometry. The Ra radionuclide 228Ra was also identified and counted.
Past testing for water quality by state, federal, and local agencies has found that higher levels of the radium isotopes 226 and 228 are present in the Cambrian-Ordovician aquifer compared with most other aquifers in the U.S. Moreover, in some locations those levels exceed the EPA maximum contaminant level (MCL) for radium. This research was also the first to measure 224Ra, which has no EPA MCL although it adds radioactivity to water because its risks to human health are lower than the other isotopes of radium. 224Ra levels were almost equal to levels of 228Ra, but generally less than levels of 226Ra.
“Ra-224 is not regulated by the U.S. EPA, although some States have promulgated their own regulations for Ra-224 knowing that it occurs at high concentrations in certain source waters,” explains Stackelberg. “Ra-224 is a short-lived radionuclide, having a ½-life of about 3.5 days, and is more difficult to analyze than Ra-226 or Ra-228. Its toxicity is less than that of Ra-226 or Ra-228. Findings from our study indicate that Ra-224 is contributing radioactivity in amounts similar to that of Ra-228 in water from the Cambrian-Ordovician aquifer system. If this water were untreated and delivered to consumers within 3.5 days of withdrawal there could be a small additional exposure to radioactivity due to the presence of Ra-224. But the risk from this would be less than that from either Ra-226 or Ra-228.”
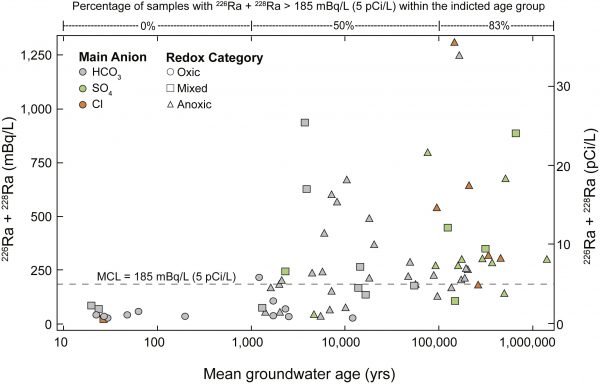
Combined Ra concentrations increase as water types change and become increasingly mineralized and anoxic with increasing groundwater age. (Credit: Authors, USGS)
Using the results
It’s important to identify the conditions that cause high levels of radium in water from the aquifer, because then it’s possible to help water utilities and resource managers prioritize resources for monitoring activities, alternative water resource development, and public education programs. When radium isotopes are the problem, various monitoring activities are recommended.
“Water from any newly installed or previously existing public-supply well is required by the U.S. EPA to be analyzed for radium,” explains Stackelberg. “Depending on the concentration of radium measured, the well is then put on a schedule for compliance monitoring. The higher the radium measurement, the more frequently the well is required to be monitored. A brief description of these monitoring requirements is available online here.”
Water managers can use the results from this study to identify the parts of the Cambrian-Ordovician aquifer system where radium concentrations most frequently exceed the U.S. EPA MCL.
“Knowing the conditions that promote elevated concentrations of radium in the Cambrian-Ordovician aquifer system – and where those conditions are most prevalent – allows water managers to (1) more effectively anticipate monitoring needs and (2) design effective and efficient monitoring networks that focus on the most important constituents associated with elevated concentrations of radium,” adds Stackelberg.
Various types of alternative water resource development are possible, once heightened risks are known. If a water purveyor knows that a potential source of drinking water for the public contains high concentrations of radium, they will have to determine if (1) it is feasible to treat that water supply to lower the radium concentration, (2) the water can be blended with other sources of water which are low in radium, or (3) alternative sources of water are available that have lower radium concentrations. Alternative sources of water could include water from other aquifer systems and/or water from nearby surface-water supplies.
“The answers to these questions will depend on the location of the water supplier,” Stackelberg expounds. “The Cambrian-Ordovician aquifer system continues to be used as a source of public and domestic drinking-water supplies—even though it’s well known that radium can occur at high concentrations—because alternative sources are not always available or adequate. In these instances, public-supply water from the Cambrian-Ordovician that has high radium is blended with other sources and/or treated to lower radium concentrations to less than the MCL.”
There is a public education component here too. Making the public aware that water from the Cambrian-Ordovician aquifer system can contain high concentrations of radium is important, because it may affect the cost of safe, clean drinking water—costs that may be passed on to the consumer. This information is also important to owners of domestic wells because they are solely responsible for the quality of their own drinking-water supplies.
“Knowing the conditions that are conducive to elevated concentrations of radium—and where these conditions are most likely to exist—will inform domestic-well owners as to whether or not they should consider having their water supply tested for radium,” Stackelberg comments; if someone tests their water and finds radium, treatment and removal are possible. “The most common treatment processes are ion exchange, lime softening, and reverse osmosis. These processes can remove most of the radium in water. More information on treating water to remove radium is available from the U.S. EPA and local and State health agencies.”

Radium in Groundwater Study Area – Cambrian-Ordovician Aquifer. (Credit: Authors, USGS)
Top image: Water type becomes increasingly mineralized (SO4 or Cl dominated) and total Ra increases where the C-O aquifer system is regionally confined by the Maquoketa Shale. (Credit: Authors, USGS)











0 comments